Radovi naših znanstvenika (19)
Disorder at the Chiral Cα Center and Room-Temperature Solid-State cis−trans Isomerization; Synthesis and Structural Characterization of Copper(II) Complexes with D-allo,L-Isoleucine
Crystal Growth Design 2018, 18, 5138−5154.
Autori: Jelena Pejić, Darko Vušak, Gábor Szalontai, Biserka Prugovečki, Draginja Mrvoš-Sermek, Dubravka Matković-Čalogović, Jasmina Sabolović
Sažetak rada
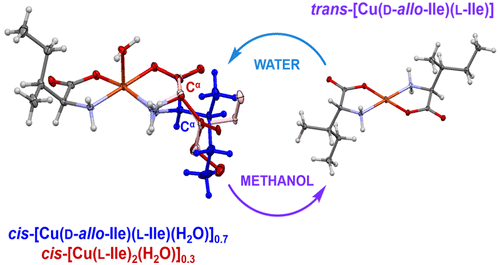
Four new complexes of copper(II) with d-allo,l-isoleucine were prepared by solution and mechanochemical synthesis: three complexes of aqua cis isomers, cis-[Cu(d-allo-Ile)2(H2O)] (2), cis-[Cu(d-allo-Ile)(l-Ile)(H2O)]0.7[Cu(l-Ile)2(H2O)]0.3 (3), cis-[Cu(d-allo-Ile)2(H2O)]0.75[Cu(d-allo-Ile)(l-Ile)(H2O)]0.25 (4), and anhydrous trans-[Cu(d-allo-Ile)(l-Ile)] (5). The previously characterized orthorhombic cis-[Cu(l-Ile)2(H2O)] (1a) cocrystallized with 3 and either 2 or 4, depending on the synthetic conditions. In 2, a disorder of the side chain was found, while 3 and 4 had a rare positional disorder of two diastereomers, namely, d-allo-Ile and l-Ile, resulting in two positions of the Cα chiral center in their crystal structures. The complexes were analyzed by X-ray diffraction methods, solid-state NMR spectroscopy, and density functional theory calculated 13C and 1H Fermi contact shifts. Room-temperature solid-state cis-to-trans isomerization of Cu(d-allo-Ile)(l-Ile) occurred by liquid-assisted grinding (LAG) of 3 with methanol, by aging in methanol vapor, and by soaking 3 in methanol. The latter was a fast process. The isomerization also happened in the 140−160 °C temperature interval. Reverse trans-to-cis isomerization occurred by LAG of 5 with water and very slowly by aging in water vapor. Low activation energy for the Cu(d-allo-Ile)(l-Ile) cis−trans isomerization in the solid state could explain why subtle changes in the experimental conditions affected the final product.
Rad je dostupan na https://pubs.acs.org/doi/10.1021/acs.cgd.8b00589
