Radovi naših znanstvenika (1)

PGLa-H tandem-repeat peptides active against multidrug resistant clinical bacterial isolates
Rončević T, Gajski G, Ilić N, Goić-Barišić I, Tonkić M, Zoranić L, Simunić J, Benincasa M, Mijaković M, Tossi A, Juretić D
Biochim Biophys Acta. 2017 Feb;1859(2):228-237. doi: 10.1016/j.bbamem.2016.11.011
Sažetak rada:
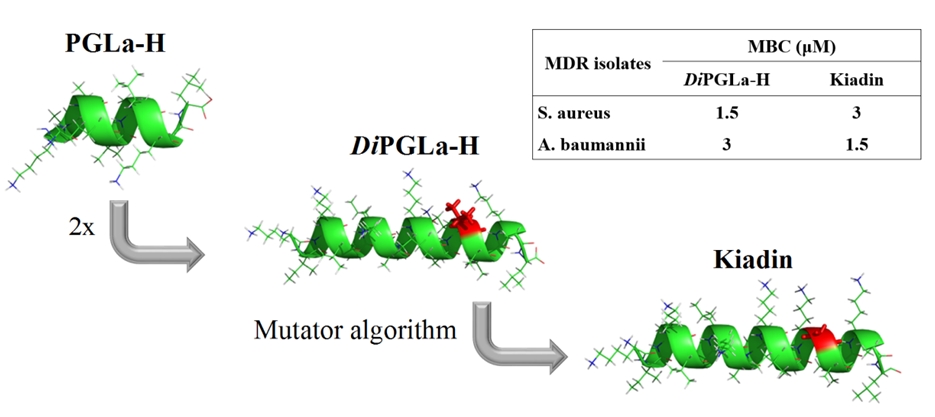
Antimicrobial peptides (AMPs) are promising candidates for new antibiotic classes but often display an unacceptably high toxicity towards human cells. A naturally produced C-terminal fragment of PGLa, named PGLa-H, has been reported to have a very low haemolytic activity while maintaining a moderate antibacterial activity. A sequential tandem repeat of this fragment, diPGLa-H, was designed, as well as an analogue with a Val to Gly substitution at a key position. These peptides showed markedly improved in vitro bacteriostatic and bactericidal activity against both reference strains and multidrug resistant clinical isolates of Gram-negative and Gram-positive pathogens, with generally low toxicity for human cells. The glycine substitution analogue, kiadin, had a slightly better antibacterial activity and reduced haemolytic activity, which may correlate with an increased flexibility of its helical structure, as deduced using molecular dynamics simulations. These peptides may serve as useful lead compounds for developing anti-infective agents against resistant Gram-negative and Gram-positive species.
Grafički sažetak:
Rad je dostupan na: http://www.sciencedirect.com/science/article/pii/S0005273616303790
